Lecture 21: Kinetics of Steelmaking, Slag Properties, and Process Overview
1. Correction: Dicalcium Silicate Formation Equation
At the beginning of the lecture, a correction is made to the stoichiometric representation of the lime-dissolution reaction discussed in the previous lecture.
Corrected Chemical Equation:
-
Reactants: Solid lime () reacts with liquid iron silicate (, known as fayalite/ferrous silicate). Note that ferrous silicate contains 2 moles of for every 1 mole of .
-
Products: Liquid “calciowüstite” ( in equimolar proportions) and solid dicalcium silicate (, containing 2 moles of per mole of ).
2. Kinetics of Steelmaking Reactions
Steelmaking involves multiphase, heterogeneous reactions (gas-metal, slag-metal). The overall rate of processes like decarburization, desulfurization, or dephosphorization is a combination of two phenomena:
-
Mass Transport: Movement of species via diffusion and convection to the reaction interface.
-
Chemical Reaction: Breaking and rearrangement of atomic bonds at the interface.
A. Rate Control Mechanisms
At steelmaking temperatures (), chemical reaction rates obey the Arrhenius rate law, meaning they are extremely fast due to exponential temperature dependence.
-
Exception: Certain reactions, like nitrogen dissolution from air or specific gasification reactions, may be chemically sluggish.
-
General Rule: Chemical reactions approach equilibrium almost instantaneously. Therefore, interfacial concentrations are dictated purely by thermodynamics (equilibrium).
Rate Expressions:
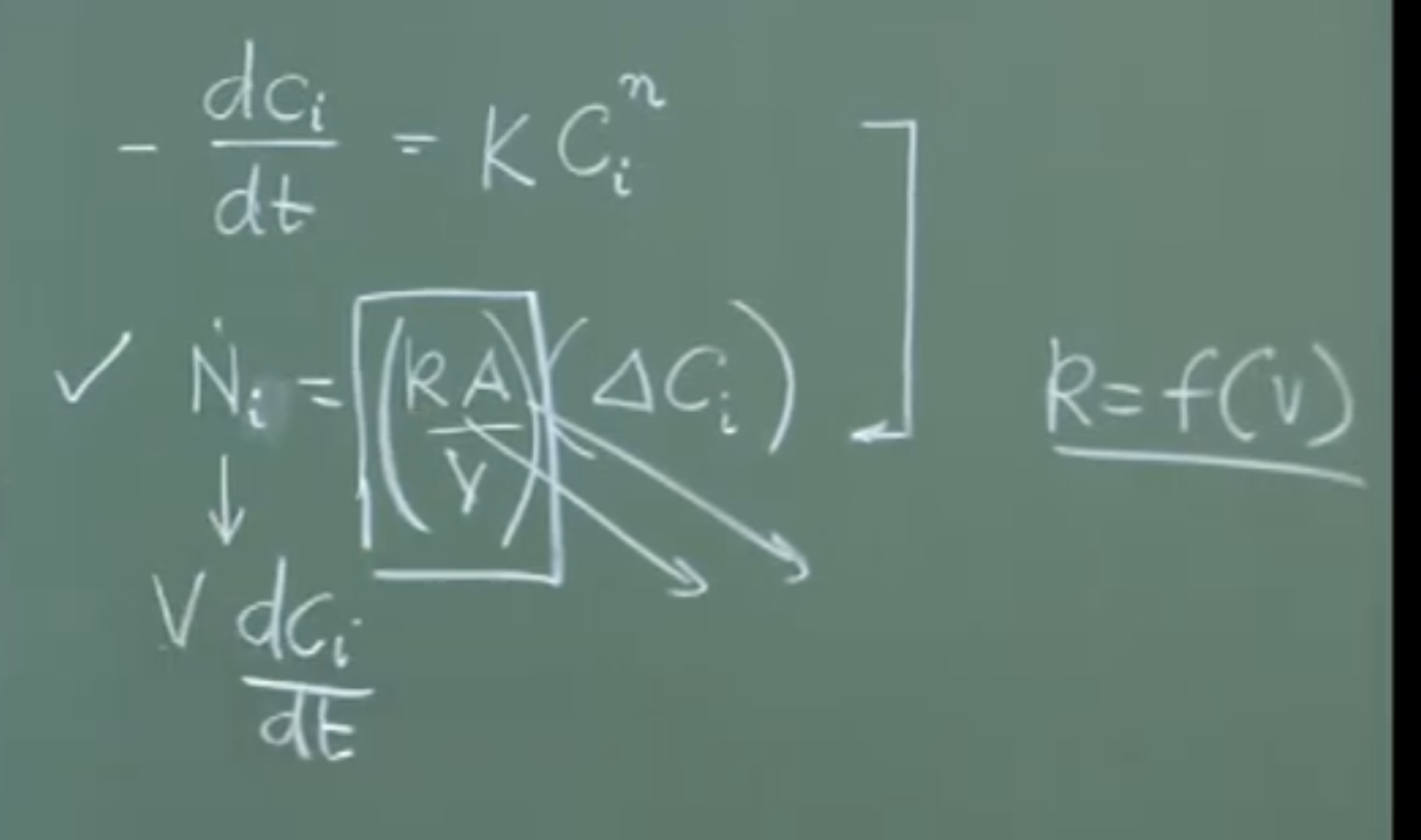
-
Chemical Rate:
(where = reaction rate constant, = reaction order)
-
Mass Transfer Rate:
(where = convective mass transfer coefficient in , = interfacial area in , = concentration gradient in )
Dimensional analysis: (mass removal rate).
Instructor Notes: Transport Control
Because chemical reactions are near-instantaneous, steelmaking kinetics are overwhelmingly Transport Controlled (specifically, Convective Mass Transfer). The rate of refining is not limited by chemistry, but by how fast you can physically push the reactants to the slag-metal or gas-metal boundary.
B. The Role of Stirring
To maximize the mass transfer rate (), you must maximize and . Both are achieved through intense stirring.
-
Increasing : Stirring increases fluid velocity, which directly increases the mass transfer coefficient.
-
Increasing (Interfacial Area): In a stagnant bath, the interfacial area is just the cross-sectional area of the vessel (). With vigorous stirring, the interface becomes wavy, and phases entrain into one another (like an emulsion of oil and water). This expands the active reaction area exponentially.
3. Chemistry and Properties of Steelmaking Slags
Steelmaking slags are highly dynamic and predominantly Quaternary Systems made of four primary oxides:
-
+ ( combined)
-
()
-
()
-
Trace impurities: Phosphorus, sulfur, etc.
A. Basicity Definitions
Basicity is a critical chemical parameter dictating the slag’s capacity to absorb impurities (like sulfide and phosphate capacities). Depending on precision needs, different definitions are used in the industry:
-
Simple V-Ratio:
-
Modified Basicity (All basics over all acids):
-
Weighted Basicity (Accounting for relative acidic/basic strengths):
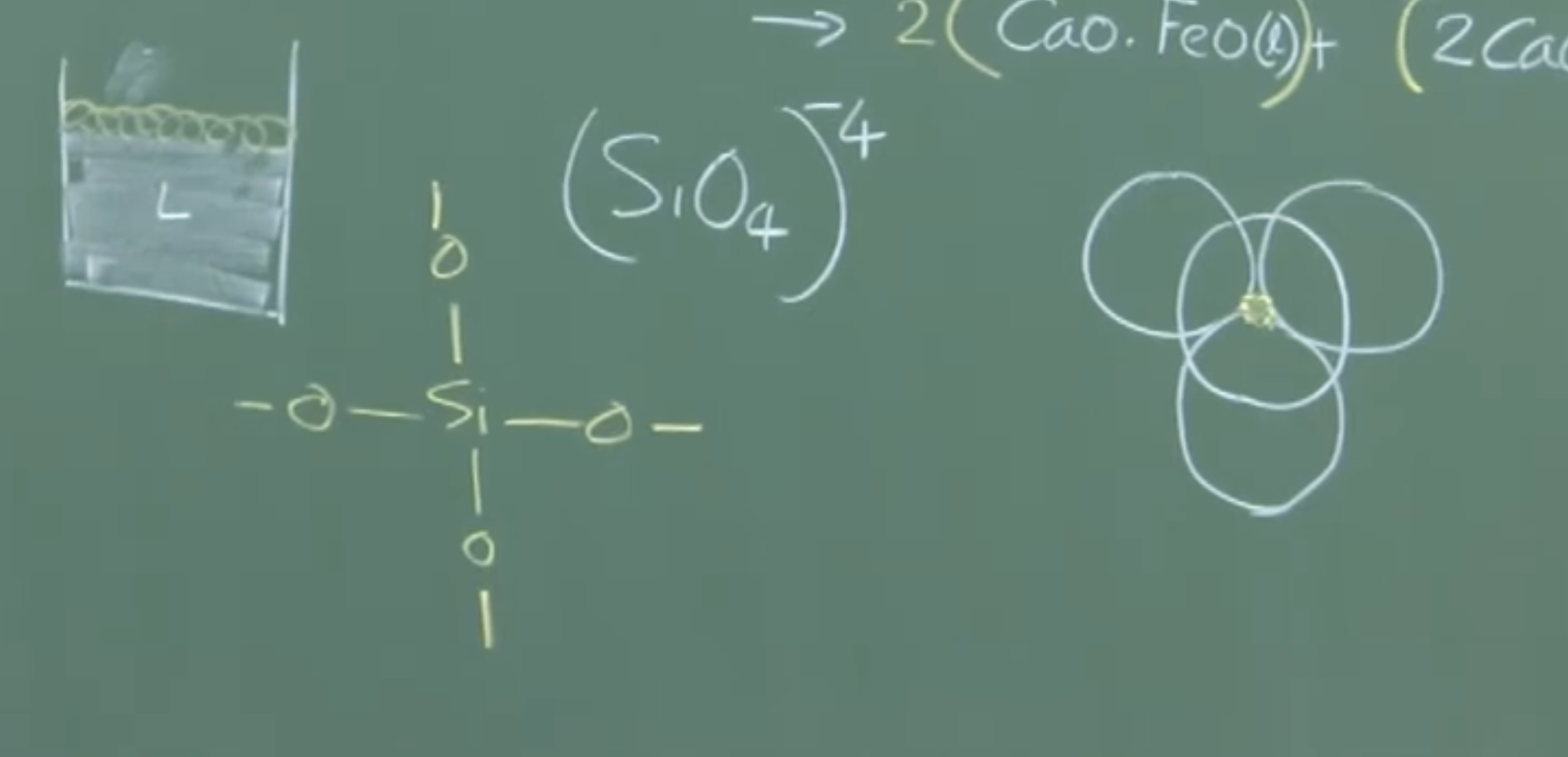
B. Silicate Network and Slag Fluidity
-
Structure: Pure silica () forms a rigid, continuous 3D network of tetrahedra sharing oxygen bonds.
-
Disruption: When basic oxides () are added, their cations () break the bridging oxygen bonds.
-
Fluidity Threshold: When the ratio of , the silicate network is completely fragmented into discrete monomers. The slag becomes highly fluid.
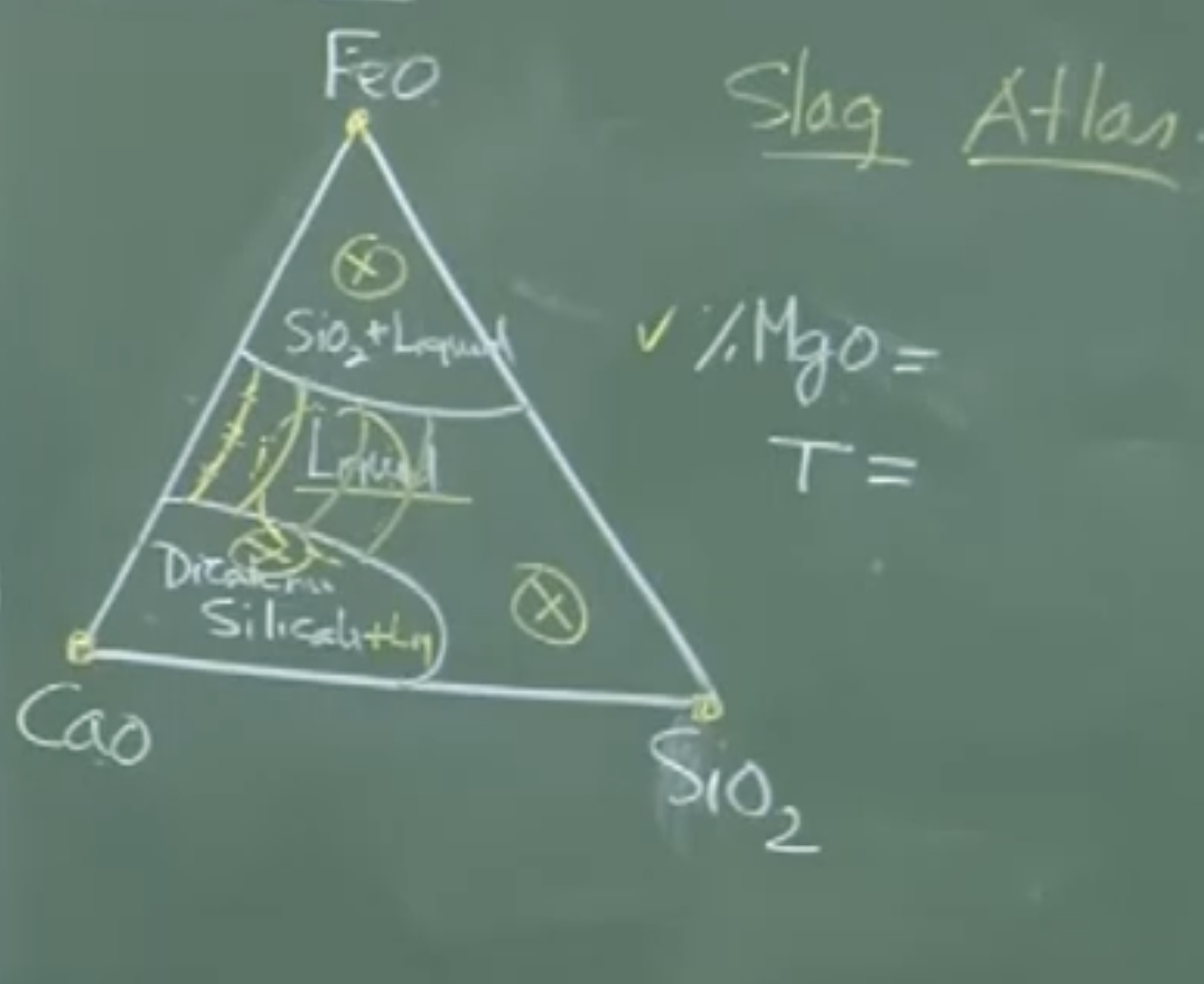
Physical Interpretation: Kinetic Slag Properties
Thermodynamics only tells you if a reaction can happen. Kinetics dictates if the slag physically allows it.
-
Fluidity/Viscosity: A highly viscous slag restricts the movement of ions (like or ), throttling the mass transfer coefficient ().
-
Melting Point: If the slag is pasty (contains undissolved solids), mass transfer crashes. Operating in a fully liquid regime is critical.
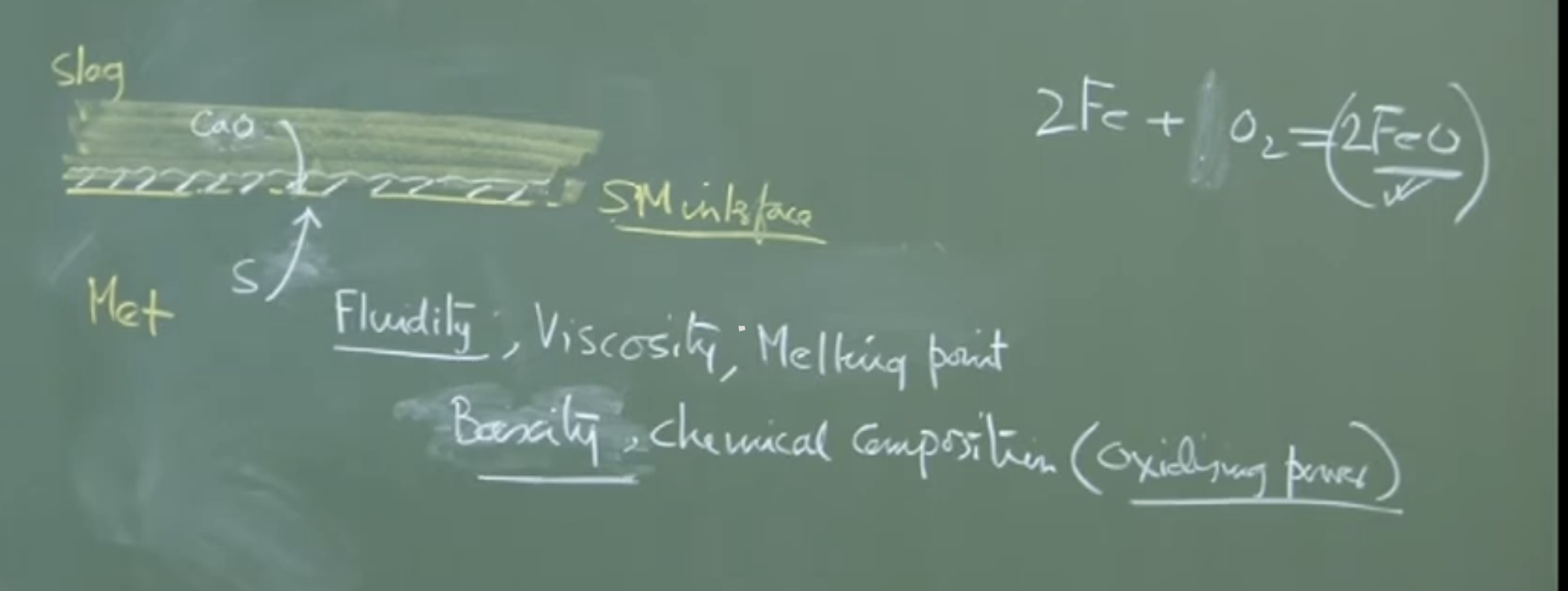
C. Chemical Potential and Ternary Phase Diagrams
-
Oxygen Potential: The oxidizing power of the slag is dictated by and represented by the chemical potential of oxygen ().
-
Activity vs. Concentration: You cannot use weight percentage directly as “activity” in thermodynamic equations. 15 wt% FeO does not mean .
-
Slag Atlas: To find actual activities, metallurgists use experimentally compiled Ternary Phase Diagrams (e.g., at a fixed and temperature).
-
These diagrams map out “Iso-activity lines” within the fully liquid phase fields.
-
They help operators steer the dynamically evolving slag composition so that it stays out of solid/pasty phase fields (like the dicalcium silicate field) and remains fully liquid.
-
4. Introduction to Modern Steelmaking Processes
Having established the thermodynamic and kinetic fundamentals, the course transitions to actual plant processes.
A. Global Steel Production Distribution
Currently, global crude steel ( billion tons/year) is dominated by two routes:
-
Basic Oxygen Steelmaking (BOS / BOF): of global production.
-
Electric Arc Furnace (EAF): of global production.
(Note: Acidic steelmaking is virtually extinct and will not be covered).
Metallic Feedstock Distribution:
-
Blast Furnace Hot Metal:
-
Scrap Steel:
-
DRI / Alternative Iron (Corex, Midrex, etc.):
B. Process Scale Comparison
| Feature | Basic Oxygen Furnace (BOF) | Electric Arc Furnace (EAF) |
|---|---|---|
| Primary Feed | Hot Metal (Liquid Pig Iron) | Steel Scrap (Solid) |
| Plant Type | Integrated Steel Plants (Giant scale) | Mini-mills (Specialty/regional scale) |
| Converter Size | 100 to 320 Tons per heat | 30 to 50 Tons per heat |
| Refining Load | Heavy (Needs to remove C, Si, P, S) | Light (Scrap is already purified steel) |
C. Historical Context: The Birth of the LD Process
-
The 20-Year Delay: Sir Henry Bessemer knew pure oxygen would make superior steel compared to air, but commercial pure oxygen wasn’t available until 1932. Even then, commercial oxygen steelmaking didn’t happen until 1952.
-
The Engineering Challenge: Why the wait? When pure is injected into a carbon/silicon-rich melt, the local heat generation is so extreme that it melts any submerged injection lances, tuyeres, and the surrounding refractory bricks within minutes.
-
The Breakthrough (1952): In the Austrian cities of Linz and Donawitz (LD), engineers discovered that the lance did not need to be submerged. By positioning a water-cooled lance above the bath and blowing oxygen down at supersonic speeds (Mach 1.5 to 2.5), the jet penetrated the slag/metal interface, creating massive internal emulsions and rapid refining without melting the lance. This birthed the modern LD / BOF process.