Lecture Notes: Steelmaking Reactions – Dephosphorization and Kinetics
1. Introduction to Dephosphorization
Dephosphorization refers to the removal of phosphorus from the hot metal. During the ironmaking stage (in the blast furnace), nearly all the phosphorus from the charge material (iron ore) is reduced and joins the molten metal phase.
-
Initial Phosphorus Content: Depending on the ore quality, hot metal typically contains 0.1% to 0.15% phosphorus by weight.
-
Target Phosphorus Content: For high-quality steel intended for critical applications, the target phosphorus content is strictly controlled to 30 ppm (0.003 wt%) or even as low as 10 ppm.
-
Note on Terminology: Just as sulfur removal is abbreviated as De-S, phosphorus elimination is denoted as De-P.
2. Thermodynamics of Dephosphorization
A. Chemical Reactions
The removal of phosphorus is inherently a slag-metal interface reaction that occurs in an oxidizing and basic environment.
Molecular Form of the Reaction:
Ionic Form of the Reaction:
B. Equilibrium Constant ()
Assuming chemical equilibrium between the metal and the slag, the equilibrium constant for the ionic reaction is given by:
Where:
-
= Activity of phosphate ion in the slag.
-
= Henrian activities of dissolved phosphorus and oxygen in the melt (which are roughly equal to their weight percentages at standard state).
-
= Activity of oxygen ions in the slag (a measure of slag basicity).
C. Equilibrium Partition Coefficient ()
We define the equilibrium partition coefficient for phosphorus, , as:
From the expression, we can infer that is proportional to:
Conceptual Explanation: Optimum Conditions for De-P
To maximize (i.e., push the maximum amount of phosphorus into the slag), the following three thermodynamic conditions must be met:
-
High Oxygen Potential (): An intense oxidizing environment (high dissolved or high in the slag) drives the reaction forward. The exponent of 5 indicates a very strong dependence on oxygen.
-
High Slag Basicity (): A high concentration of basic oxides (like ) captures the oxidized phosphorus to form stable calcium phosphate.
-
Low Temperature: The dephosphorization reaction is strongly exothermic. According to Le Chatelier’s principle, lowering the temperature increases and shifts the equilibrium to the right (product side).
Instructor Note: Contrast this with Desulfurization (De-S), which requires a highly basic but reducing environment. Because De-P requires highly oxidizing conditions, De-P is ideally suited for the steelmaking converter, whereas De-S is better handled in the blast furnace or via hot metal pretreatment.
3. Phosphate Capacity and Empirical Relationships
Just as basic slags possess a “Sulfide Capacity” (), they also possess a “Phosphate Capacity” (), which quantifies the slag’s intrinsic ability to absorb phosphorus.
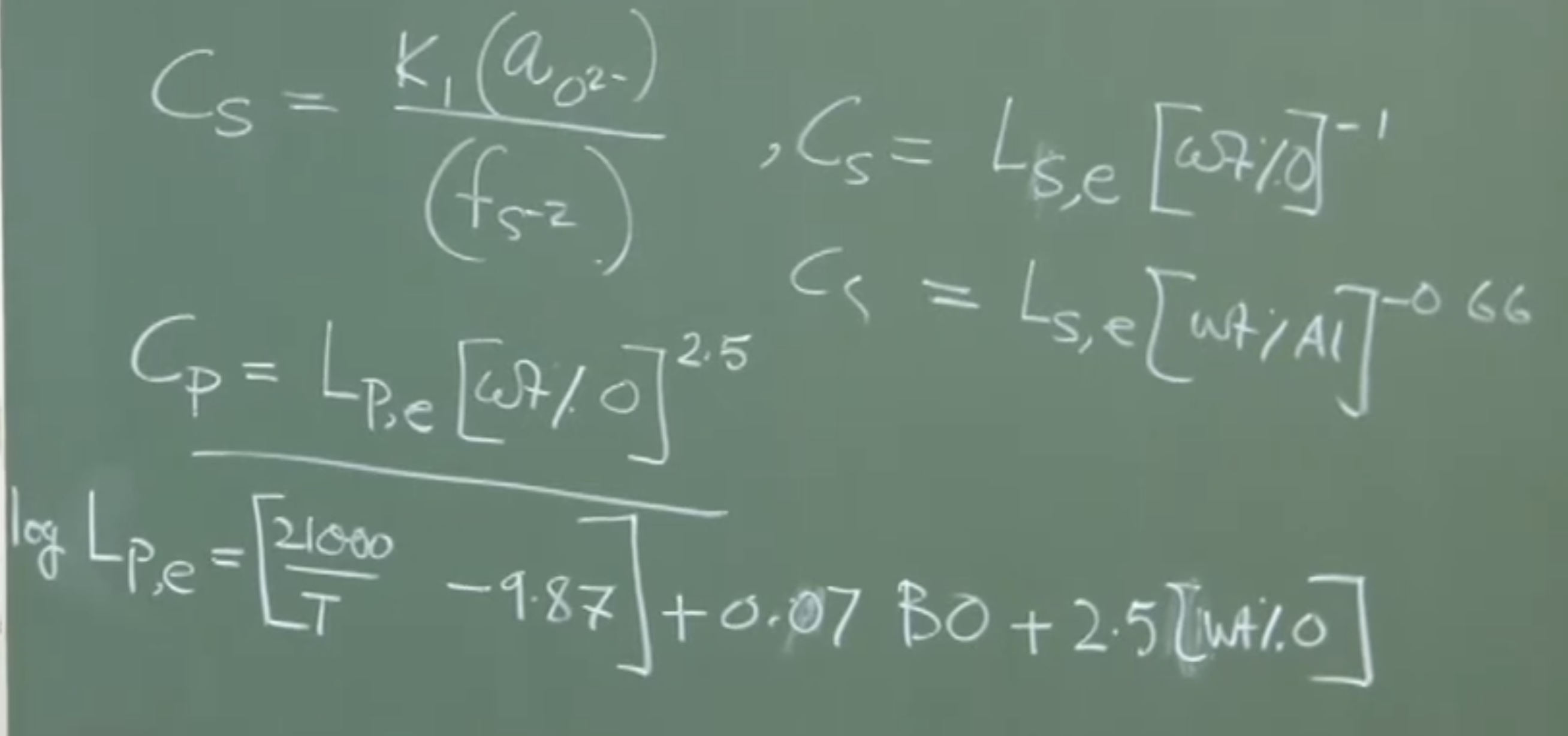 Empirical Equation for :
Empirical Equation for :
Researchers have established a quantitative relationship for the partition coefficient as a function of temperature, basicity, and oxygen:
Where is a basicity parameter defined as:
(Note: Calcium fluoride () is historically used to fluidize slags, but its use is declining due to environmental restrictions on fluoride contamination).
Visual Extraction: Graphical Relationships
Graph 1: Effect of Temperature and Basicity on
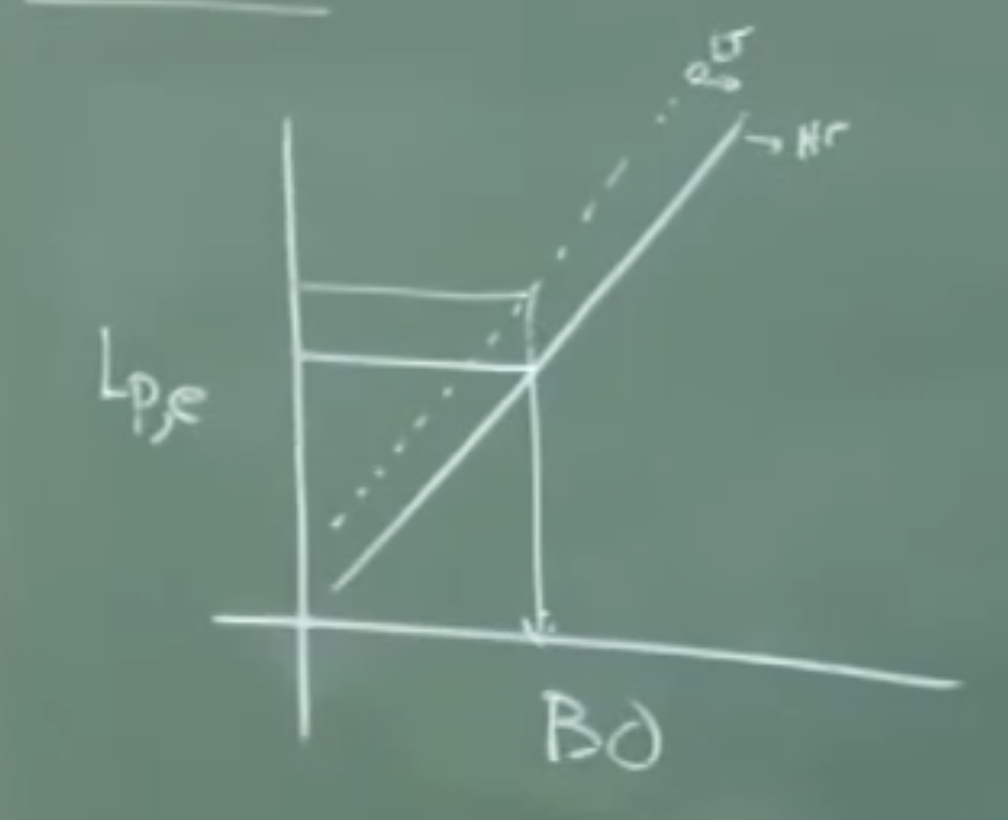
log(L_P)
^
| / Line A (e.g., 1600 °C) -> Lower T
| /
| / / Line B (e.g., 1690 °C) -> Higher T
| / /
| / /
| / /
| / /
|______________________ V_o (Basicity)
Physical Interpretation: For any given basicity ( or ), the partition coefficient () is higher on Line A than Line B. Because the De-P reaction is exothermic, Line A must represent the lower temperature condition.
Graph 2: Effect of Content on Dephosphorization
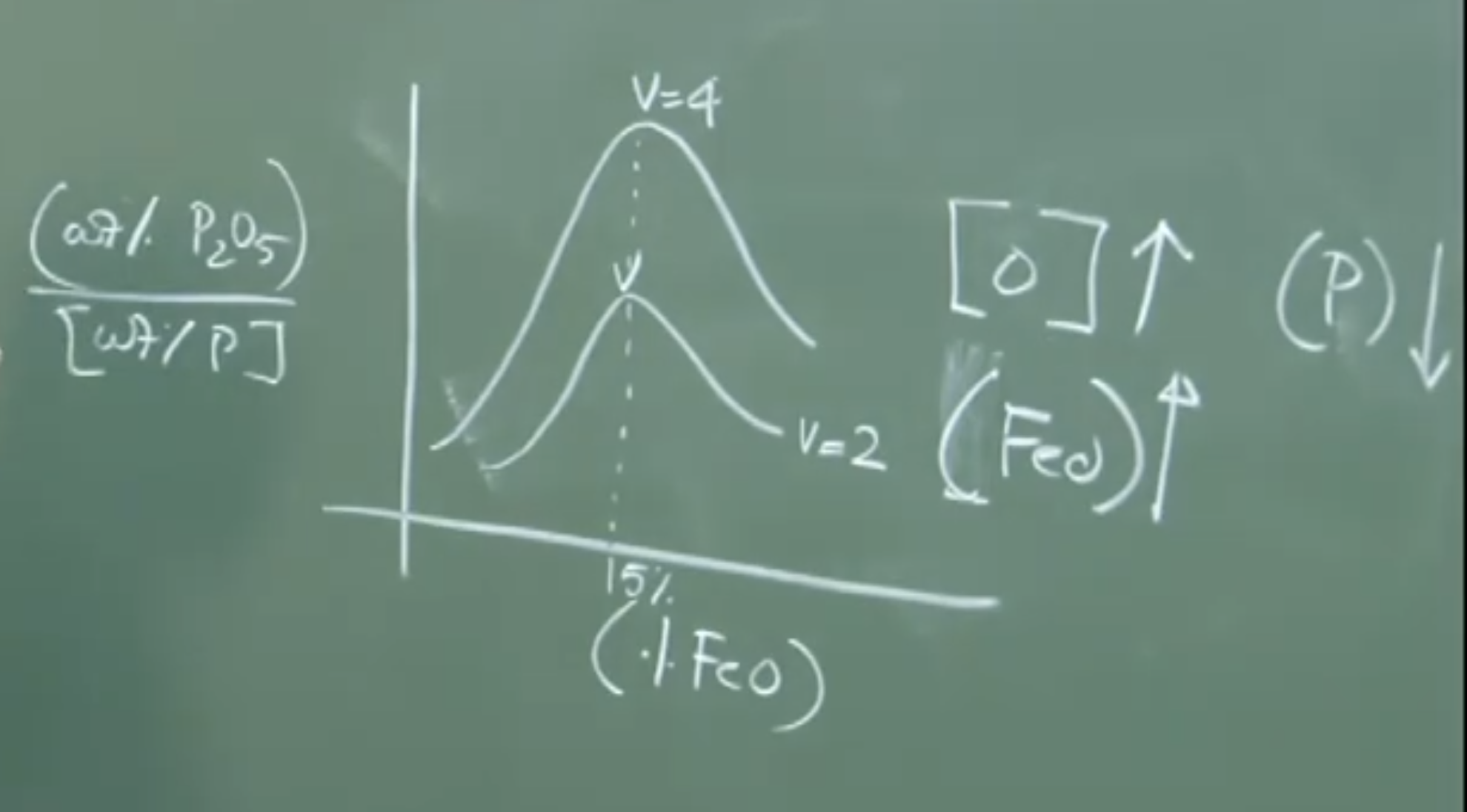
(wt% P2O5 / wt% [P])
^
| ___
| / \
| / \
| / \
| / \
| / \
|__/_______________\_______ wt% (FeO) in slag
~ 15%
Physical Interpretation: De-P initially increases as increases (higher oxygen potential). However, beyond , the dephosphorization efficiency suddenly drops. To understand why, we must examine the kinetics of slag formation.
4. Slag Formation and Lime Dissolution Kinetics
The basicity of the slag doesn’t magically reach ; lime () must physically dissolve into the liquid slag.
-
Early in the blow, silicon and iron oxidize rapidly, forming a relatively low-melting acidic/neutral liquid slag: .
-
Solid lime () particles are added. They are lighter than the molten metal and float in this initial liquid slag phase.
The Dicalcium Silicate Problem:
When the solid reacts with the liquid iron-silicate slag, an intermediate chemical reaction occurs at the solid-liquid interface:
 Important Remarks: Dicalcium silicate precipitates as a fine solid powder exactly at the boundary of the lime particle. This solid precipitate completely coats the unreacted lime particle with an impervious layer.
Important Remarks: Dicalcium silicate precipitates as a fine solid powder exactly at the boundary of the lime particle. This solid precipitate completely coats the unreacted lime particle with an impervious layer.
Because the is trapped inside a solid shell, it can no longer dissolve into the bulk slag. Consequently, the bulk slag basicity stops increasing. This is why De-P drops at very high initial —the cannot dissolve to capture the phosphorus!
Phosphorus Reversion
Towards the end of the steelmaking blow, carbon levels are extremely low. Oxygen instead reacts heavily with iron to form , releasing a massive amount of heat.
-
The sudden spike in bath temperature (exothermic iron oxidation).
-
Because De-P is favored at low temperatures, this extreme heat forces the phosphate ions to dissociate.
-
Phosphorus moves from the slag back into the metal (Phosphorus Reversion).
-
Conclusion: Precise “End-Point Control” (stopping the oxygen blow at the exact right moment) is critical to prevent reversion.
5. Kinetics of Steelmaking Reactions
Steelmaking involves multiphase, heterogeneous reactions (gas-metal (eg. , slag-metal (eg. , solid-liquid). These reactions only occur at the phase boundaries as heterogeneous .
The Five Steps of a Heterogeneous Reaction (Example: De-Sulphurization)
Consider the reaction:
To complete this reaction, the following sequence must occur in series:
-
Mass Transfer 1: Transport of dissolved from the bulk metal to the slag-metal interface.
-
Mass Transfer 2: Transport of basic from the bulk slag to the interface.
-
Chemical Reaction: Breaking and rearranging of bonds precisely at the interface to form and release .
-
Mass Transfer 3: Transport of the product away from the interface into the bulk slag.
-
Mass Transfer 4: Transport of the byproduct away from the interface into the bulk metal.
Rate-Limiting Step
Because steelmaking operates at exceptionally high temperatures (), the Arrhenius dependency of chemical reaction rates means Step 3 (chemical bond rearrangement) occurs almost instantaneously.
-
Therefore, the chemical reaction is almost never the bottleneck.
-
The rate-limiting steps are the Mass Transfer steps (1, 2, 4, or 5). Specifically, diffusion across the boundary layers (not the well-mixed bulk) slows the process down.
-
Role of Stirring: This is exactly why pneumatic oxygen steelmaking is infinitely faster than the older Open Hearth process. Vigorous stirring physically destroys the boundary layers and rapidly continuously supplies fresh reactants to the interface.
V2: Detailed
Lecture 20: Steelmaking Reactions – Dephosphorization and Kinetics
I. Introduction to Dephosphorization (De-P)
Dephosphorization refers to the removal of phosphorus from hot metal during oxidizing refining (steelmaking).
-
Initial State: In the blast furnace, almost all phosphorus from the charge materials (iron ore) is reduced and dissolves into the hot metal. Depending on the ore grade, hot metal typically contains 0.1% to 0.15% phosphorus by weight.
-
Target State: For critical applications, modern primary and secondary steelmaking aims for extremely low final phosphorus levels, typically 30 ppm (0.003 wt%) or even as low as 10 ppm.
II. Thermodynamics of Dephosphorization
A. Chemical Reaction Equations
Phosphorus removal is a slag-metal interfacial reaction that requires both an oxidizing and a basic environment.
Molecular Form:
Ionic Form:
Instructor Note: The reaction is strictly heterogeneous. Phosphorus and oxygen are dissolved in the metal phase (, ), while the calcium oxide and resulting phosphates exist in the slag phase (, ).
B. Equilibrium Constant ()
Assuming slag-metal equilibrium, the equilibrium constant for the ionic reaction can be written as:
Where:
-
= Activity of the phosphate ion in the slag.
-
= Henrian activities of dissolved phosphorus and oxygen in the melt (approximately equal to their weight percentages in a 1 wt% standard state).
-
= Activity of oxygen ions in the slag (a direct measure of slag basicity).
C. Equilibrium Partition Coefficient ()
To measure the efficiency of phosphorus removal, we define the Equilibrium Partition Coefficient ():
From the expression, we can see that is highly dependent on oxygen potential and basicity:
D. Optimum Conditions for Dephosphorization
To maximize (drive phosphorus into the slag), three essential thermodynamic conditions must be met:
-
High Oxygen Potential (): Synonymous with high dissolved in the melt or high in the slag. The exponent of 5 indicates a massive dependence on oxidizing conditions.
-
High Slag Basicity (): Abundant basic oxides (like ) are required to capture the oxidized phosphorus and stabilize it in the slag.
-
Relatively Low Temperature: The De-P reaction is highly exothermic. Lower temperatures favor the forward reaction (increasing ).
Conceptual Explanation: De-P vs. De-S
Desulfurization (De-S) requires a highly basic but reducing environment. Because De-P requires a highly oxidizing environment, it is best suited for the steelmaking converter, whereas De-S is better handled in the blast furnace or via hot metal pretreatment.
III. Phosphate Capacity and Empirical Models
Just as slags have a “Sulfide Capacity,” they also possess a Phosphate Capacity—a thermodynamic parameter indicating the slag’s intrinsic ability to absorb phosphorus.
A. Empirical Equation for
Extensive industrial experiments yield empirical relationships to calculate . A representative equation is:
-
Temperature (): The positive term confirms that lower temperatures increase .
-
Basicity Parameter (): A common industrial definition is . (Note: usage is currently facing worldwide environmental bans).
B. Phosphorus Reversion
-
The Phenomenon: Towards the end of the oxygen blow, carbon is heavily depleted. Oxygen begins to heavily oxidize iron, which is an intensely exothermic reaction.
-
The Consequence: The sudden, drastic increase in bath temperature causes the calcium phosphate in the slag to thermodynamically dissociate. Phosphorus leaves the slag and re-enters the metal. This is known as Phosphorus Reversion.
-
Solution: Strict “End-Point Control” (stopping the oxygen blow at the precise target carbon/temperature) is mandatory to prevent reversion.
IV. Slag Formation and Lime Dissolution Kinetics
Thermodynamics dictates that high and high increase De-P. However, kinetically, maxes out at around 15 wt% FeO and then drops. This is due to the physical mechanics of slag formation.
A. The Dicalcium Silicate Barrier
-
Initial Slag: Early in the blow, oxidized silicon and iron form a liquid iron-silicate slag ().
-
Lime Addition: Solid lime () particles are added. They float in the liquid iron-silicate slag.
-
Interfacial Reaction: At the solid-liquid boundary, they react:
-
The Choke Point: The dicalcium silicate precipitates as a solid, fine powder directly on the surface of the lime particle. This forms an impervious coating that prevents the inner unreacted lime from dissolving into the bulk slag.
Physical Interpretation: If the content is too high (), this solid layer rapidly forms and sinters, locking the away. Even though you added enough lime for a basicity of 4, the effective basicity of the liquid slag remains low because the lime cannot dissolve. Therefore, De-P drops.
V. Kinetics of Steelmaking Reactions
Are these reactions fast enough for industrial production? Steelmaking relies heavily on heterogeneous, multi-phase reactions (gas-metal, slag-metal, solid-liquid).
A. The Five Steps of a Heterogeneous Reaction
Using Desulfurization as an example (), the reaction must occur at the exact phase boundary and consists of five sequential steps:
-
Mass Transfer 1: Transport of reactant from the bulk metal across the boundary layer to the interface.
-
Mass Transfer 2: Transport of reactant from the bulk slag to the interface.
-
Chemical Reaction: Breaking and rearranging chemical bonds at the interface to form .
-
Mass Transfer 3: Transport of product away from the interface into the bulk slag.
-
Mass Transfer 4: Transport of byproduct away from the interface into the bulk metal.
B. The Rate-Limiting Step
-
Chemical Kinetics are Fast: Because steelmaking occurs at elevated temperatures (), the Arrhenius equation dictates that the chemical reaction step (Step 3) happens almost instantaneously.
-
Mass Transfer is Slow: Therefore, the overall speed of the reaction is entirely governed by the slowest step in the sequence—which is universally Mass Transfer (diffusion across the boundary layers).
Physical Interpretation: The Tea and Sugar Analogy
If you put sugar in a cup of tea and do not stir it, the sugar will just sit at the bottom. The chemical potential for dissolution is there, but the mass transfer is too slow. The moment you use a spoon to stir, the sugar dissolves rapidly.
Conclusion: In steelmaking, vigorous stirring/agitation (like injecting oxygen at supersonic speeds) physically shreds the boundary layers, expediting mass transfer and making the refining reactions incredibly fast.